I’m proud to announce the publication of an article in Volume 56, Issue 3 of the Journal of Emergency Medicine describing the first documented use of Mar-Med’s Derma-Stent device on a human patient. The case was performed in the context of a randomized controlled clinical trial at Wayne State University under principal investigator Aaron Brody, MD, MPH. The now completed trial involved 60 participants comparing standard packing gauze vs Derma-Stent in various outcomes for abscess treatment. While the final manuscript on the trial is being written, The Journal has published Dr. Brody’s description of using Derma-Stent on the trial’s first enrollee.
Read the newly published clinical trial article here!
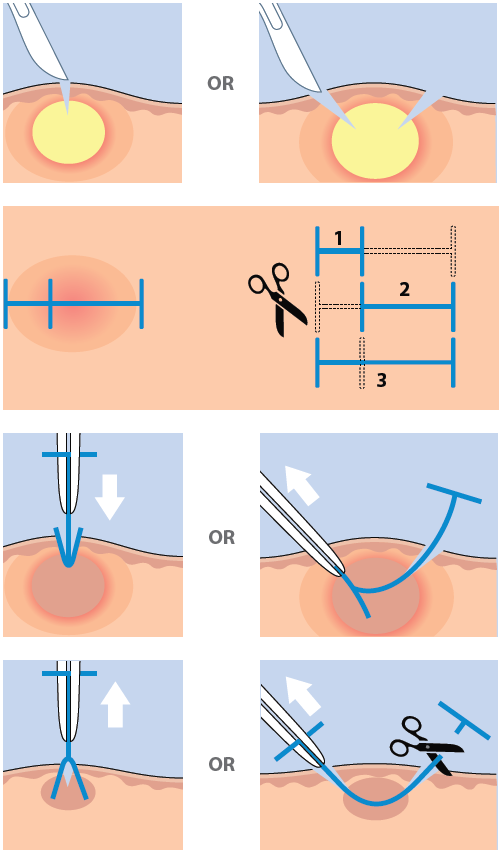
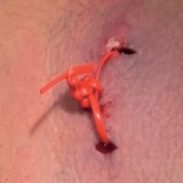
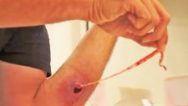
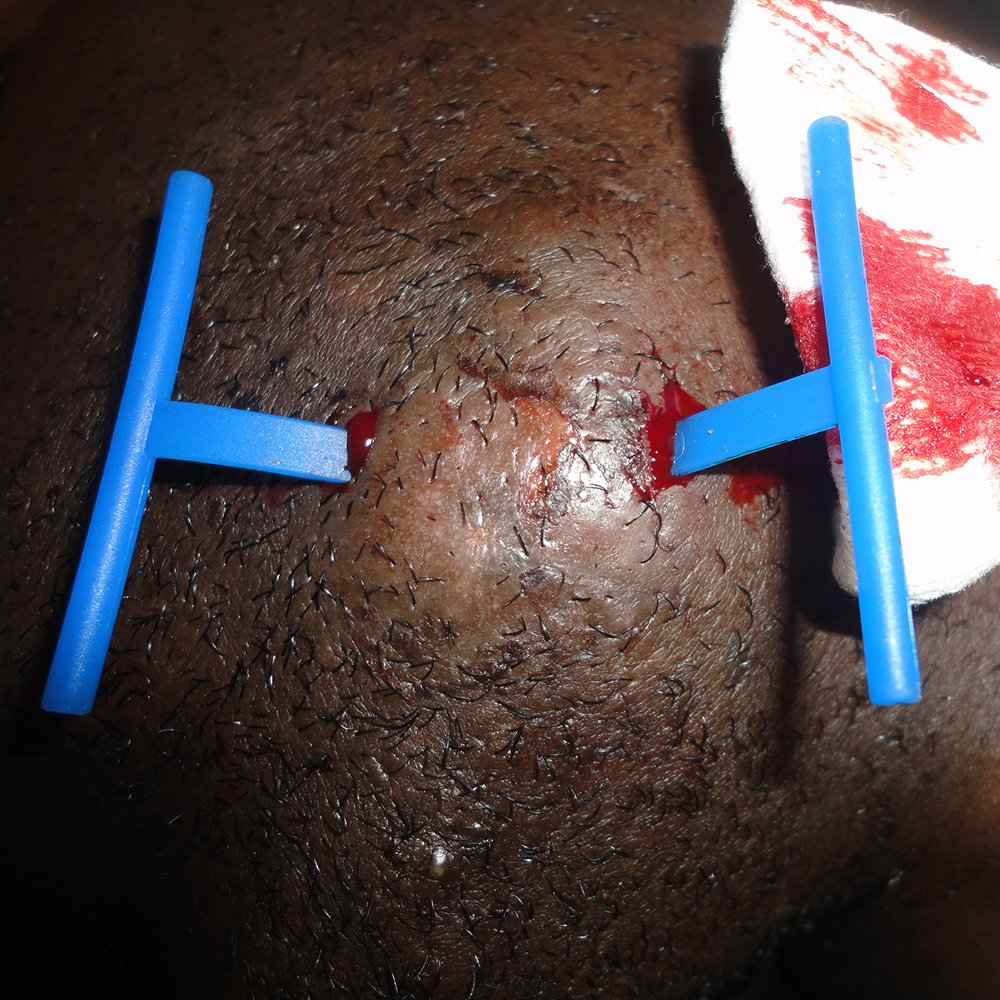
I designed Derma-Stent after speaking with numerous emergency clinicians about the changing nature of abscess treatment. It is no great wonder why the field is moving away from medieval packing gauze. It’s absorbent, uncomfortable, and often requires multiple clinician visits. The improvised “loop drain” method (vessel loop, Penrose, or other elastic) is gaining attention and for good reason, but also has disadvantages which limit its adoption. My goal with Derma-Stent is to help retire packing gauze by providing a simple, low cost device specifically for cutaneous drainage. Modeled after the concept of a price tag fastener (see below), Derma-Stent can be customized to stick into 1 or 2 incisions without tying clumsy knots or sutures. And soft, bio-compatible silicone makes Derma-Stent comfortable and easy for clinicians and patients to manage.
Don’t take my word for it. Feel free to read about Dr. Aaron Brody’s take by visiting our new clinical trial page here, where you can find excerpts from the article as well as the full download until May 11. While I’m listed as an author on the article, I’d like to note that I had no discretion over its content. We also encourage you to try Derma-Stent for yourself by requesting samples at our website via www.marmed.com/sample-request.
Derma-Stent Tutorial: